Edwards INTUITY Elite
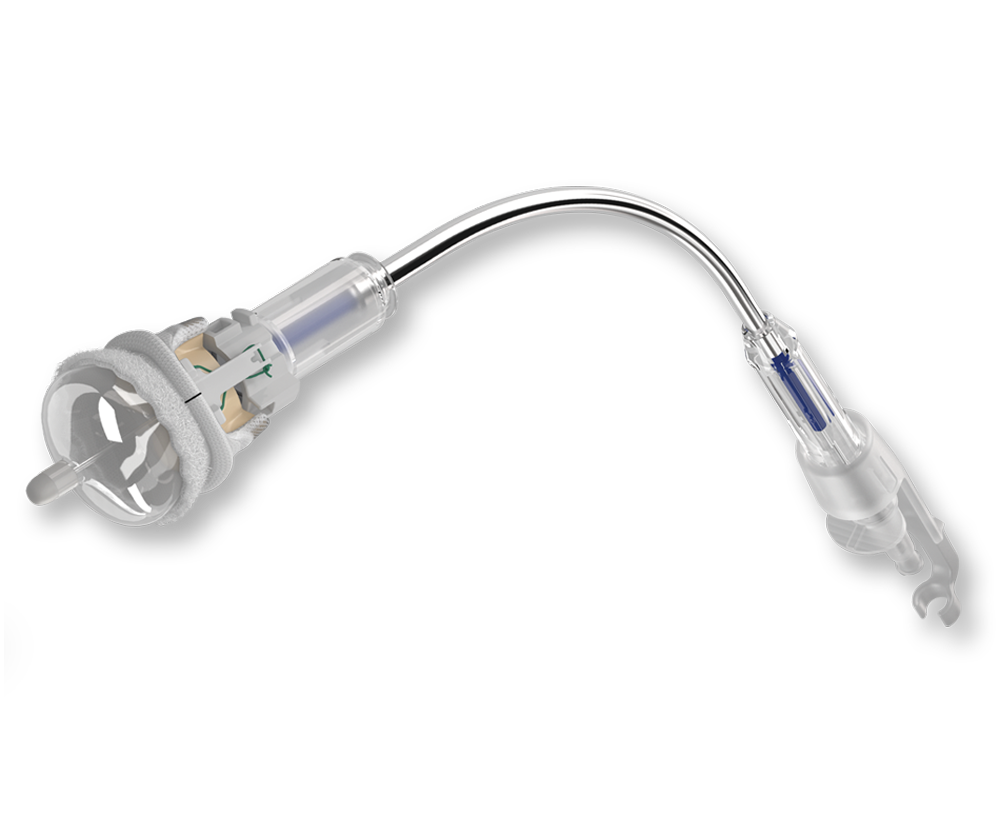

The EDWARDS INTUITY Elite valve system is designed to achieve three important goals simultaneously:
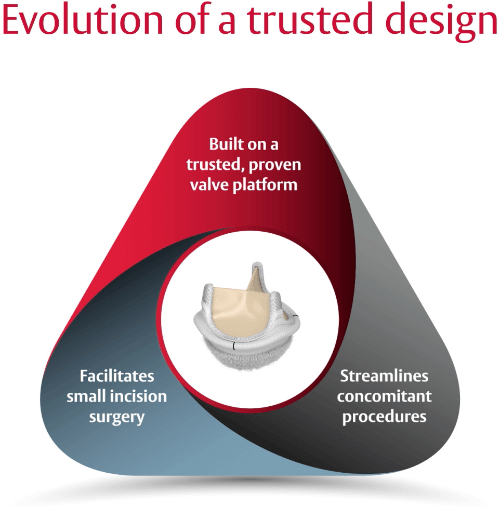
The EDWARDS INTUITY Elite valve system represents our commitment to surgeons and patients in heart valve therapy.
We have combined our proven pericardial valve technology with our innovations in transcatheter heart valves to create a surgical valve designed to streamline procedures and facilitate smaller incision surgery. We believe more efficient, less invasive procedures can provide significant benefits, both during the procedure and after.
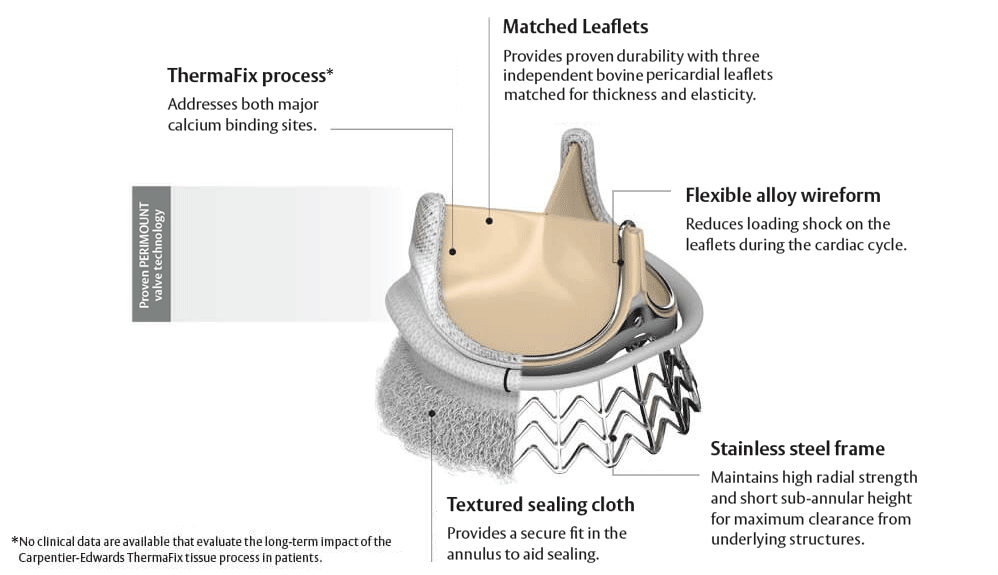
Built on a trusted, proven valve platform
Designed for durability. Created to last.
The EDWARDS INTUITY Elite valve system combines our proven pericardial valve technology with our innovations in transcatheter heart valves.
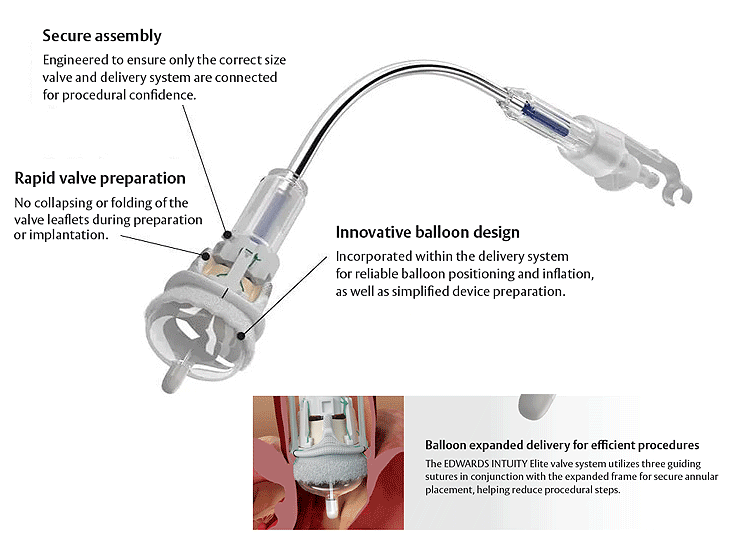
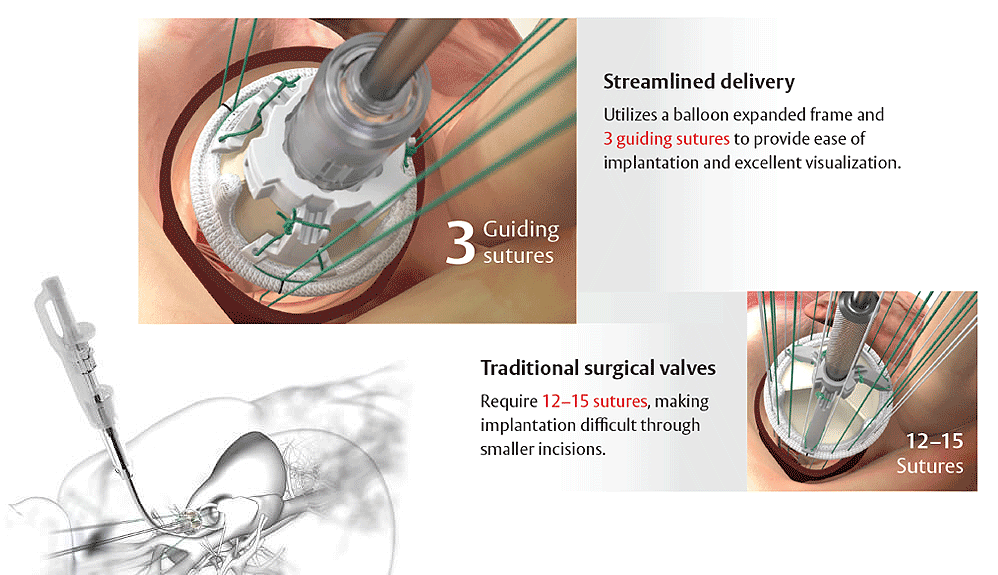
Provides rapid deployment for streamlined procedures
Streamlined implantation. Because time is precious.
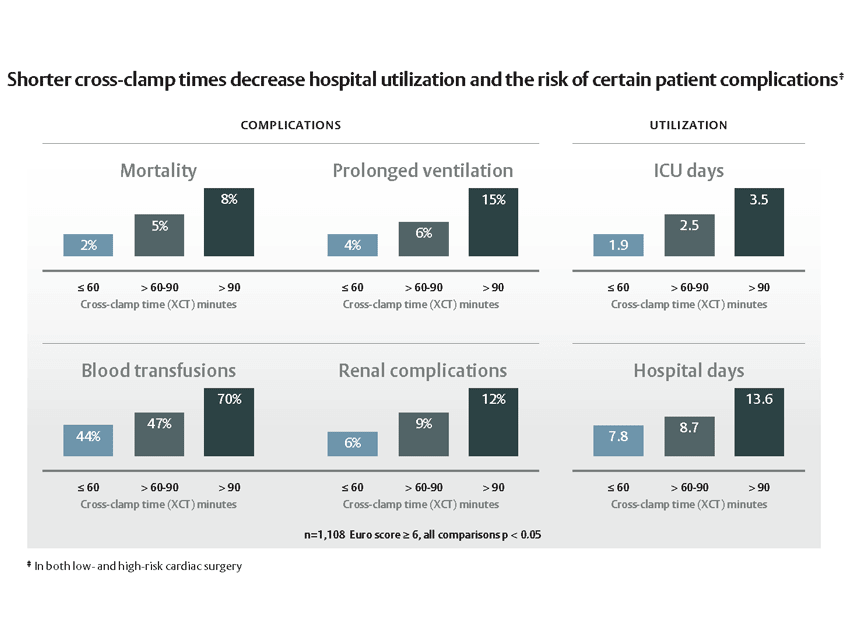
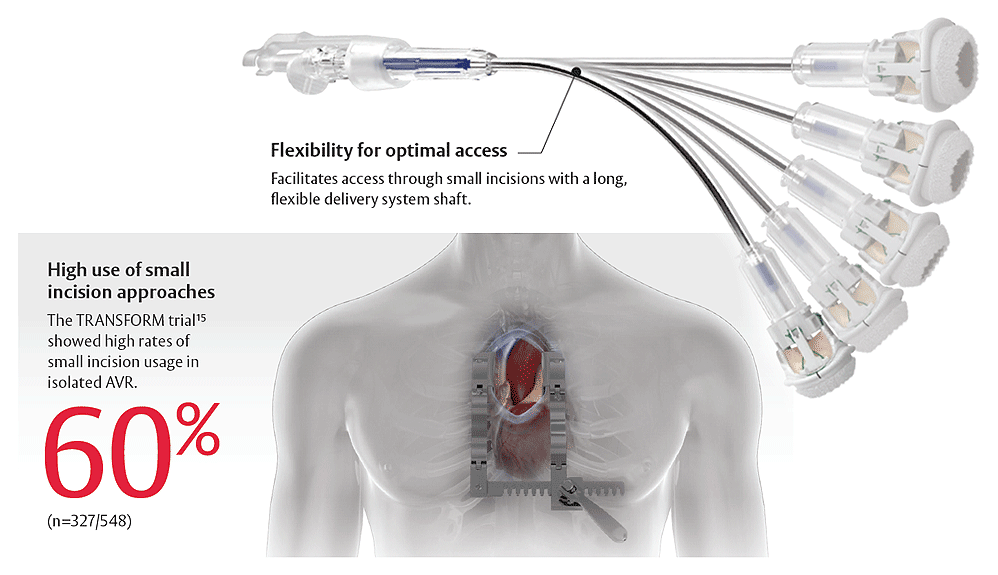
Facilitates small incision surgery
Empowering multiple approaches. Progress through access.
The EDWARDS INTUITY Elite valve system is designed to enhance the ease of implantation through small incisions using three guiding sutures.
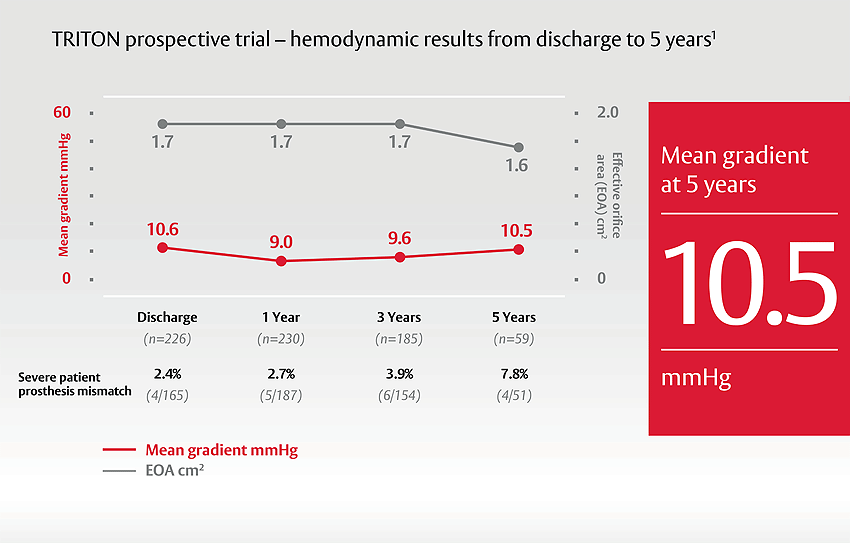
Excellent hemodynamic performance
Consistent excellent EOA and low gradients up to 5 years were observed during the TRITON study.
Preparation animation
Procedure animation
References
- Laufer G, Haverich A, Andreas M, et al. Long-term outcomes of a rapid deployment aortic valve: data up to 5 years. Eur J Cardiothorac Surg. 2017;52(2):281-287
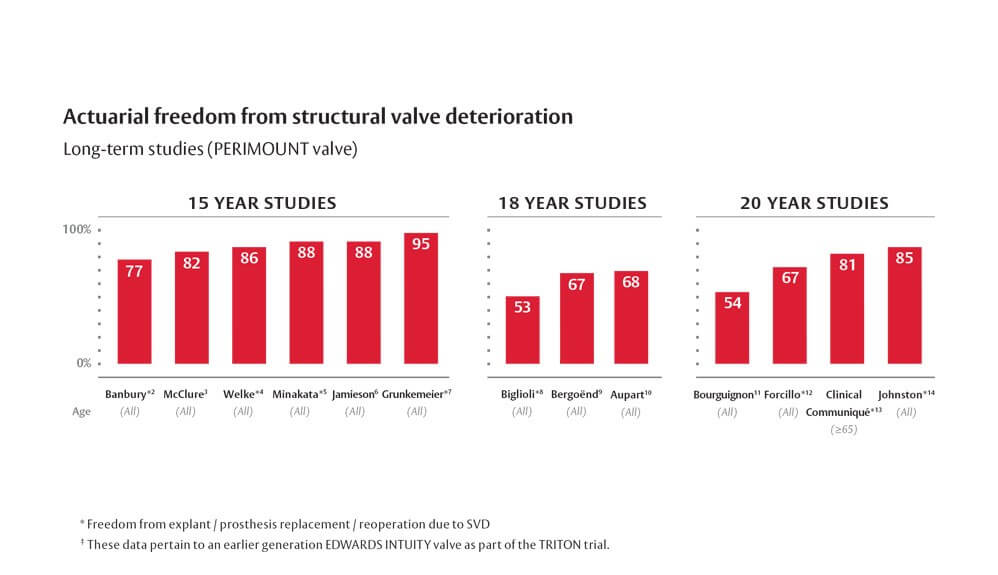
- Banbury MK, Cosgrove DM III, White JA, et al. Age and Valve Size Effect on the Long-term Durability of the Carpentier-Edwards Aortic Pericardial Bioprosthesis. Ann Thorac Surg. 2001;72(3):753-757. (Cohort size = 267, mean age = 65 ± 12 yrs. Number at risk for Explant for SVD at last follow-up not reported)
- McClure RS, Narayanasamy N, Wiegerinck E, et al. Late Outcomes for Aortic Valve Replacement with the Carpentier-Edwards Pericardial Bioprosthesis: Up to 17-year Follow-up in 1,000 Patients. Ann Thorac Surg. 2010;89(5):1410-1416. (Cohort size = 1,000, mean age = 74.1 ± 0.29 yrs. Number at risk for SVD at last follow-up not reported)
- Welke KF, Wu Y, Grunkemeier GL, et al. Long-term results after Carpentier-Edwards pericardial aortic valve implantation, with attention to the impact of age. The Heart Surgery Forum. 2011;14(3):E160-165. (Cohort size = 2,168, mean age = 73.1 ± 10.8. Number at risk at last follow-up not reported)
- Minakata K, Tanaka S, Okawa, Y, et al. Long-Term Outcome of the Carpentier-Edwards Pericardial Valve in the Aortic Position in Japanese Patients. Circulation Journal 2014;78:882-889. (Cohort size = 574, mean age = 71.9 yrs. Number at risk for Structural Deterioration at 15-year follow-up = 2)
- Jamieson WR, Germann E, Aupart MR, et al. 15-year Comparison of Supra-annular Porcine and PERIMOUNT Aortic Bioprostheses. Asian Cardiovasc Thorac Ann. 2006;14(3):200-205. (Cohort size = 1,430, mean age = 69.5 ± 10.4 yrs. Number at risk for SVD at last follow-up = 33)
- Grunkemeier GL, Furnary AP, Wu Y, et al. Durability of pericardial versus porcine bioprosthetic heart valves. The Journal of Thoracic and Cardiovascular Surgery. 2012;144(6):1381-1386. (Cohort size = 2,449, mean age = 72.9. Number at risk for explantation for SVD at last follow-up not reported)
- Biglioli P, Spampinato N, Cannata A, et al. Long-term outcomes of the Carpentier-Edwards pericardial valve prosthesis in the aortic position: effect of patient age. J Heart Valve Dis. 2004;13(1):S49-51. (Cohort size = 327, mean age = 67.2 ± 10.6 yrs. Number at risk for Prosthesis Replacement at last follow-up not reported)
- Bergoënd E, Aupart MR, Mirza A, et al. 20 years’ durability of Carpentier-Edwards Perimount stented pericardial aortic valve. In: Yankah CA, Weng Y, Hetzer R,eds. Aortic Root Surgery The Biological Solution. Berlin: Springer; 2010:441-451. (Cohort size = 1,857, mean age = 69.8 yrs, Number at risk for Structural Valve Deterioration at last follow-up = 36)
- Aupart MR, Mirza A, Meurisse YA, et al. Perimount Pericardial Bioprosthesis for Aortic Calcified Stenosis: 18-year Experience with 1133 Patients. J Heart Valve Dis. 2006;15(6):768-775. (Cohort size = 1,133, mean age = 72.6 yrs. Number at risk for SVD at last follow-up = 2)
- Bourguignon T, Bouquiaux-Stablo, A, Candolfi P, et al. Very Long-Term Outcomes of the Carpentier-Edwards PERIMOUNT Valve in Aortic Position. Ann Thorac Surg. 2015;99(3):831-7. (Cohort size = 2,659, mean age = 71 ± 10.4 yrs. Number at risk for explant for Structural Valve Deterioration = 28).
- Forcillo J, Pellerin M, Perrault LP, et al. Carpentier-Edwards Pericardial Valve in the Aortic Position: 25-Years Experience. Ann Thorac Surg 2013;96:486-93. (Cohort size = 2,405, mean age = 71 ± 9 yrs. Number at risk for Structural Deterioration at last follow-up = 30)
- Clinical Communiqué. Carpentier-Edwards PERIMOUNT Aortic Pericardial Bioprosthesis 20-year Results. Data on file at Edwards Lifesciences, 2003. (Cohort size = 267, mean age = 65 ± 12 yrs. For patients ≥ 65, number at risk for explant due to SVD at last follow-up = 2)
- Johnston DR, Soltesz EG, Vakil N, et al. Long-term durability of bioprosthetic aortic valves: implications from 12,569 implants. Ann Thorac Surg. 2015;99(4):1239-47. (Cohort size = 12,569, mean age = 71 ±11 yrs. Number at risk for explant for Structural Valve Deterioration at 20 year follow-up = 54).
- Barnhart GR, Accola KD, Grossi EA, et al. TRANSFORM (Multicenter Experience With Rapid Deployment Edwards INTUITY Valve System for Aortic Valve Replacement) US clinical trial: Performance of a rapid deployment aortic valve. J Thorac Cardiovasc Surg. 2017;153(2):241-251.e2.
Important safety information
EDWARDS INTUITY Elite valve system
Indications: The EDWARDS INTUITY Elite valve is indicated for the replacement of diseased, damaged or malfunctioning native or prosthetic aortic valves.
Contraindications: The EDWARDS INTUITY Elite valve is contraindicated for use in patients with pure aortic insufficiency and aneurysms of the aortic root or ascending aorta.
Warnings: Use of the EDWARDS INTUITY Elite valve system may be associated with new or worsened conduction system disturbances, which may require a permanent pacemaker implant.
Complications and Side Effects: Annulus damage, dissection or tearhemolysis, cardiac arrhythmias/conduction disturbances, congestive heart failure, endocarditis, leaflet impingement (aortic or mitral), myocardial infarction (MI), neurologic events, patient-prosthesis mismatch (PPM) (due to inappropriate sizing), structural/non-structural valve dysfunction, any of which could lead to reoperation, explantation, and death.
With the use of a bioprosthetic valve with a reduced number of sutures similar to the EDWARDS INTUITY Elite valve include: valve leakage, paravalvular (perivalvular) leak, transvalvular regurgitation, valve stenosis, valve thrombosis, valve frame distortion (from chest compression or trauma), and valve malposition, instability, dislodgement or migration/embolization.
CAUTION: Federal (USA) law restricts these devices to sale by or on the order of a physician. See instructions for use for full prescribing information.