EDWARDS’ INSPIRIS RESILIA VALVE RECEIVES FDA APPROVAL
IRVINE, Calif., July 5, 2017 - Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced it received U.S. Food and Drug Administration (FDA) approval for its INSPIRIS RESILIA aortic valve, the first in a new class of resilient heart valves.
“The INSPIRIS valve sets a new benchmark for surgical aortic valve replacement, providing patients, particularly those with active lifestyles, with a resilient tissue valve that is built on Edwards’ legacy of excellence in heart valve innovations,” said John D. Puskas, MD, MSc, FACC, FACS, professor of Cardiothoracic Surgery, Icahn School of Medicine at Mount Sinai, chairman, Department of Cardiothoracic Surgery, Mount Sinai Saint Luke's, Mount Sinai Beth Israel and Mount Sinai West (Roosevelt) and national principal investigator of the COMMENCE pivotal trial, an FDA premarket approval study.
Joseph Bavaria, MD, Roberts-Measey professor and vice chief of cardiovascular surgery at the University of Pennsylvania and the top implanter in the COMMENCE pivotal trial, said, "With the addition of VFit technology, featuring the unique expandable design feature, the INSPIRIS valve provides an ideal foundation for heart valve patients, both now and in the future."
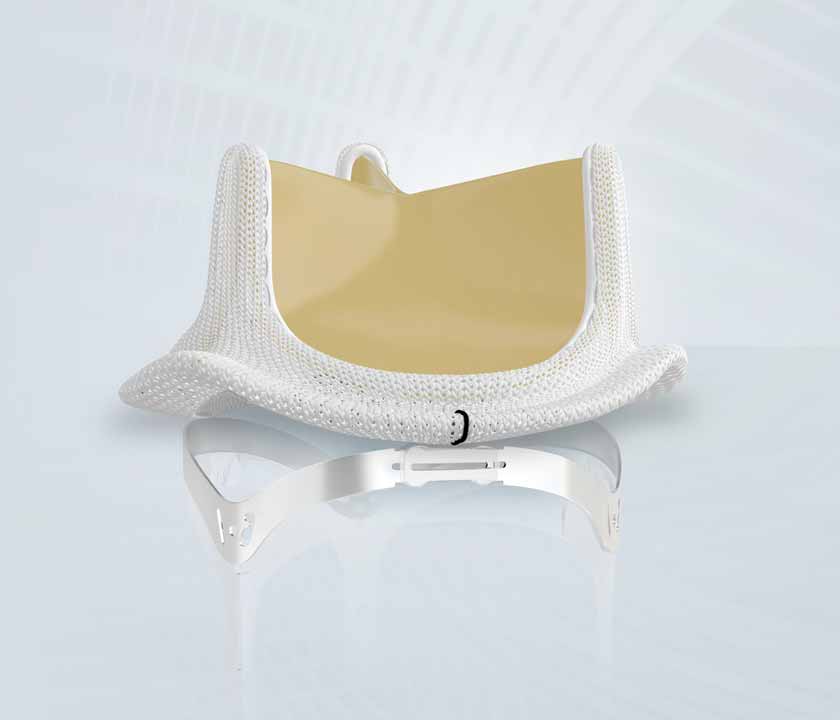
The INSPIRIS valve features three differentiating elements:
- RESILIA tissue, a breakthrough tissue technology that delivers the combination of enhanced anti-calcification properties, improved sustained hemodynamic performance and dry storage;
- A first-of-its-kind expandable frame, VFit technology also incorporates fluoroscopically visible size markers and is designed for potential future valve-in-valve procedures;
- A trusted valve platform built on the proven performance of the Carpentier-Edwards PERIMOUNT valve design, which has more than 20 years of published clinical durability.
Approval of the INSPIRIS valve is supported by the COMMENCE pivotal trial. Published in the European Journal of Cardio-Thoracic Surgery, two-year data of 689 patients from the COMMENCE trial showed no cases of structural valve deterioration, valve thrombosis or nonstructural valve dysfunction. In addition, the European RESILIA tissue feasibility study, which initiated enrollment in 2011, underscores the safety and effectiveness of the RESILIA tissue. Four-year results from the European feasibility study will be presented at the 31st European Association for Cardio-Thoracic Surgery’s (EACTS) annual meeting in October.
“Building on Edwards’ 40 years of leadership in tissue valve technology, the INSPIRIS valve featuring RESILIA tissue has been in development for more than a decade and may be uniquely suited for future valve-in-valve procedures,” said Bernard Zovighian, Edwards’ corporate vice president, surgical heart valve therapy. “In partnership with surgeons, Edwards is committed to continually developing transformative surgical innovations that benefit patients.”
Plans are underway for a full U.S. launch of the INSPIRIS valve in early 2018. The valve became commercially available in Europe earlier this year.
About Edwards Lifesciences
Edwards Lifesciences, based in Irvine, Calif., is the global leader in patient-focused medical innovations for structural heart disease, as well as critical care and surgical monitoring. Driven by a passion to help patients, the company collaborates with the world’s leading clinicians and researchers to address unmet healthcare needs, working to improve patient outcomes and enhance lives. For more information, visit www.edwards.com and follow us on Twitter @EdwardsLifesci.
This news release includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. These forward-looking statements include, but are not limited to, statements by Drs. Puskas and Bavaria and Mr. Zovighian and expectations regarding the product’s potential benefits and risks, as well as expected future innovations. Forward-looking statements are based on estimates and assumptions made by management of the company and are believed to be reasonable, though they are inherently uncertain and difficult to predict. Our forward-looking statements speak only as of the date on which they are made and we do not undertake any obligation to update any forward-looking statement to reflect events or circumstances after the date of the statement.
Forward-looking statements involve risks and uncertainties that could cause results to differ materially from those expressed or implied by the forward-looking statements based on a number of factors, including but not limited to, unanticipated outcomes of expanded and longer term clinical experience with the product, or unanticipated manufacturing, quality, development or regulatory delays or issues. These and other additional factors are detailed in the company's filings with the Securities and Exchange Commission including its Annual Report on Form 10-K for the year ended December 31, 2016. These filings, along with important safety information about our products, may be found at www.edwards.com.
Media Contact: Carol Thornton, 949-250-3472
Investor Contact: Mark Wilterding, 949-250-6826