100%
From one heart to over 500,000 lives touched.

Together, we are building a comprehensive body of evidence to drive innovation and advance patient care.
MOMENTIS Study of the MITRIS RESILIA Valve: One of the Largest Core-Lab Adjudicated Surgical Mitral Valve Replacement (MVR) Studies
Results at 1 year
An ongoing prospective, observational, post-market, single-arm, real-world evidence study in 351 North American adults with a dysfunctional native or prosthetic mitral valve who required replacement with a MITRIS RESILIA Mitral Valve.1
99.7%
Freedom from valve-related death
99.7%
Freedom from valve-related reintervention
For more details, download the clinical infographic.

Explore the clinical data
At the 1-year follow-up, the MOMENTIS study demonstrated promising results for real-world mitral valve patients with the MITRIS RESILIA mitral valve.
MOMENTIS study follow-up is ongoing and will continue through 10 years
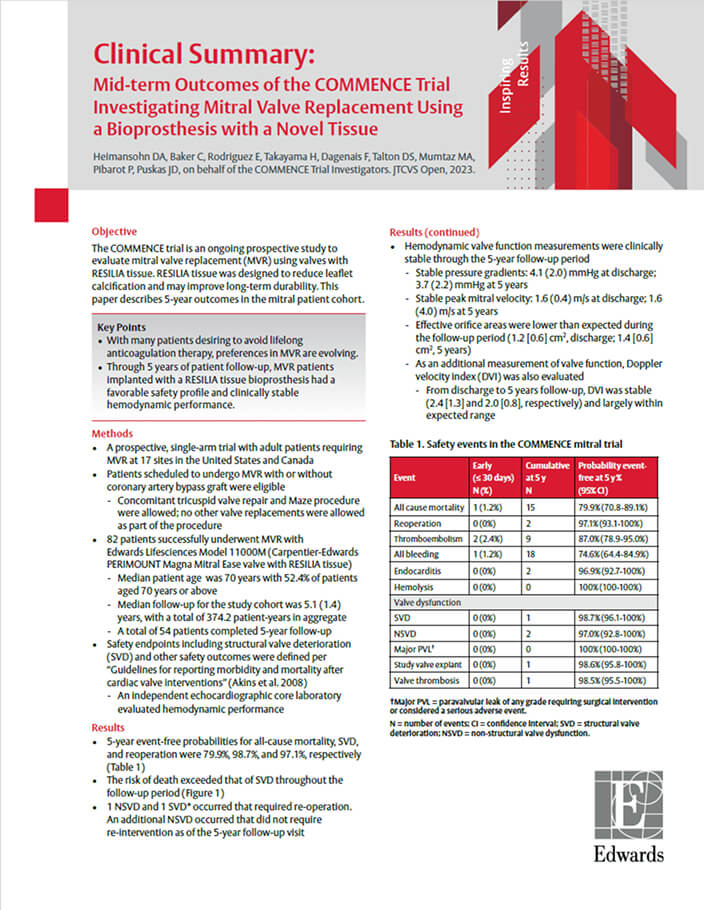
COMMENCE Mitral Trial: Early Clinical Data Supporting RESILIA Tissue in the Mitral Position
Results at 5 years
Mid-term outcomes of the ongoing COMMENCE mitral IDE trial of 82 patients who underwent MVR in this prospective, single-arm trial.2
79.9%
Freedom from all-cause mortality
98.7%
Freedom from SVD
97.1%
Freedom from reoperation
For more details, download the clinical summary.
Explore the clinical data
With a favorable safety profile and clinically stable hemodynamic performance, this study supports the use of RESILIA tissue in the mitral position with excellent durability out to 5 years.
RESILIA tissue valves in the mitral position offer excellent durability out to 5 years
References
- Ailawadi G, Heimansohn D, Accola K, et al. One-year safety and performance of a novel bovine pericardial mitral valve with advanced anti-calcification treatment. Presented at the 2026 Society of Thoracic Surgeons Annual Meeting. January 29, 2026.
- Heimansohn DA, Baker C, Rodriguez E, et al. Mid-term outcomes of the COMMENCE trial investigating mitral valve replacement using a bioprosthesis with a novel tissue. JTCVS Open. 2023;15:151-163. Published 2023 Jun 2. doi:10.1016/j.xjon.2023.05.008
- Akins CW, Miller DC, Turina MI, et al. Guidelines for reporting mortality and morbidity after cardiac valve interventions. Ann Thorac Surg. 2008;85(4):1490-1495. doi:10.1016/j.athoracsur.2007.12.082
Medical device for professional use.
Medical device for professional use.
For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
PP--EU-1509 v1.0
