From one heart to over 500,000 lives touched.

Together, we are building a comprehensive body of evidence to drive innovation and advance patient care.
EU Feasibility Study
Results at 5 years
A prospective, multicenter, single-arm trial of 133 patients who underwent surgical aortic valve replacement (SAVR).1
Freedom from all-cause mortality
Freedom from valve thrombosis
For more details, download the clinical summary.
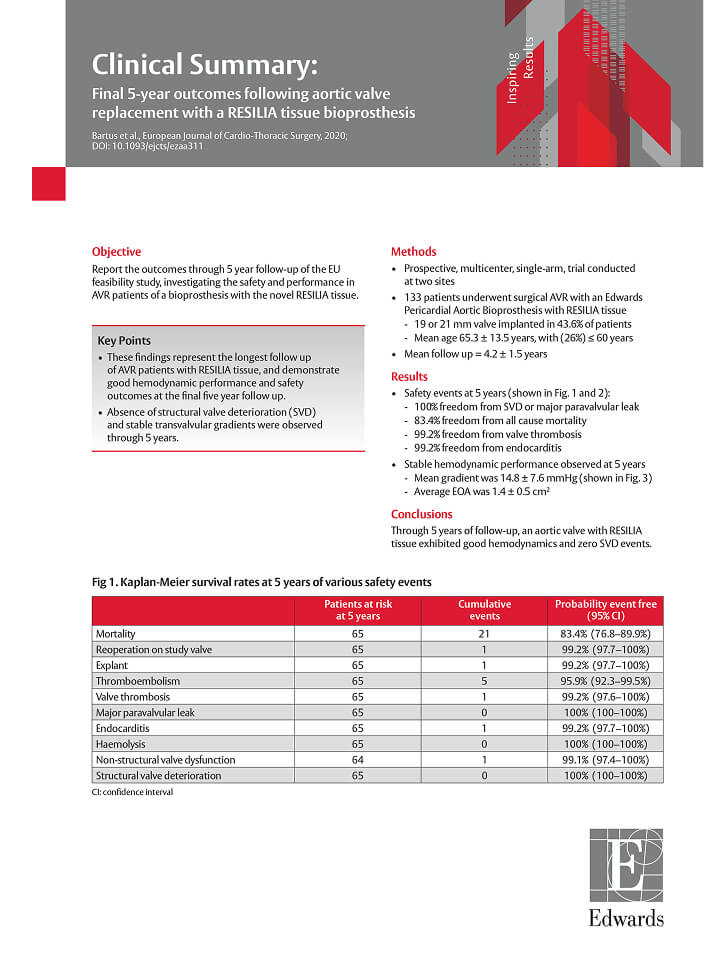
The RESILIA tissue valve demonstrated a good safety profile and excellent hemodynamic performance over 5 years of follow-up.
Preclinical Study: Pannus Formation Animal Studies
Results of 2 animal studies
Two independent studies, a 5-month study and an 8-month study, using a juvenile sheep model of mitral valve replacement with bovine pericardial tissue.2
Five-month study results
pannus growth vs 2.61 ± 2.15 mm2
Eight-month study results
pannus growth vs 0.134 ± 0.066 mm2
RESILIA tissue valves were associated with reduced pannus formation vs control valves, demonstrating the potential to improve long-term outcomes for patients.
Preclinical Study: Juvenile Sheep Study
Results
Forty-five juvenile sheep were randomized to assess an advanced tissue preservation technology. Of these, 31 survived for treatment evaluation (17 RESILIA tissue, 14 control valves with XenoLogiX treatment). Echocardiography at 1 week showed normal valve function in both groups. At 8 months, cardiac output increased significantly to the same extent in both groups (vs baseline; P <.01). The mean transvalvular pressure gradient also increased, but significantly more in the control group compared with the test group (P=.03).3
calcium content vs 6.8 ± 1.6 μg/mg
For more details, download the clinical paper
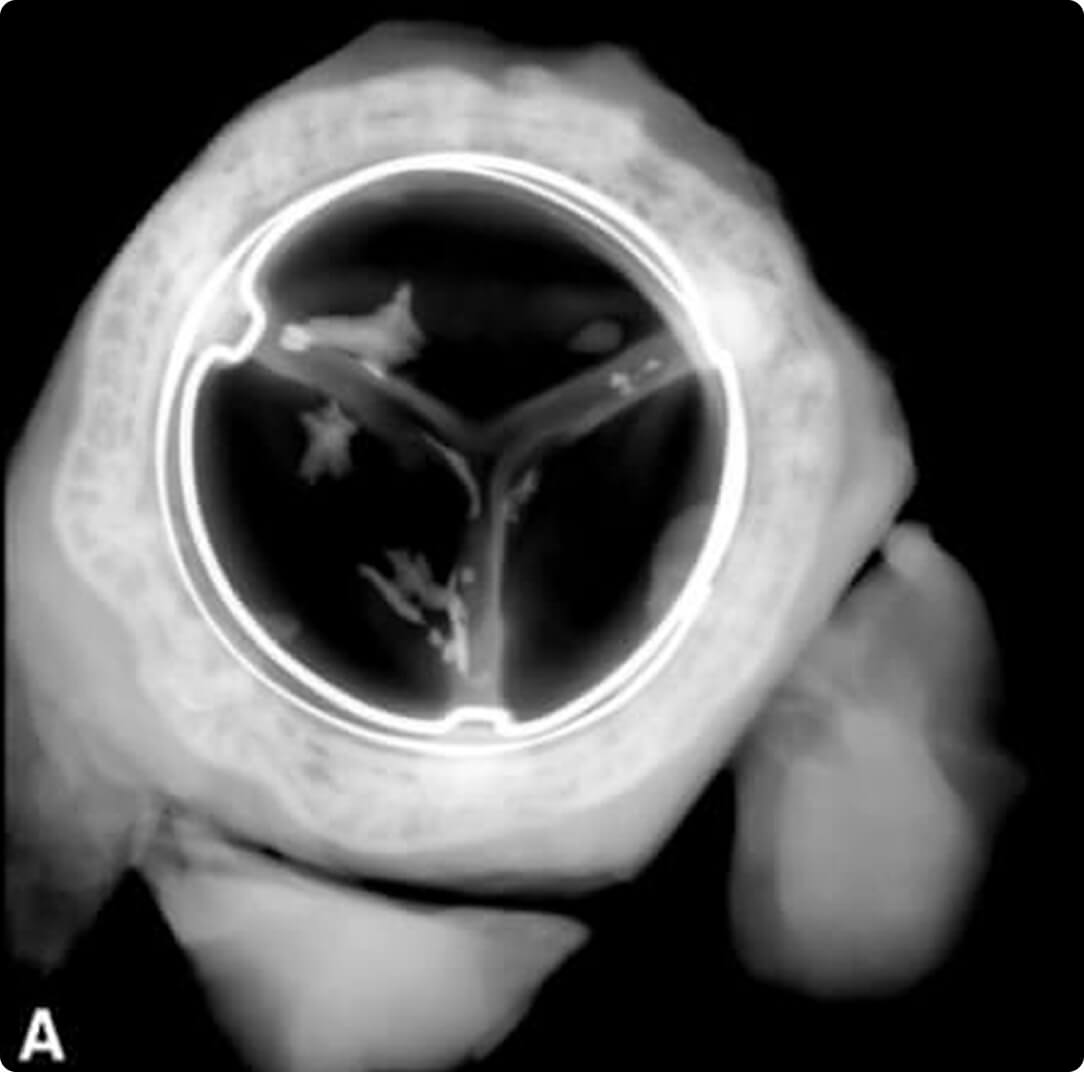
A = PERIMOUNT Tissue Valve
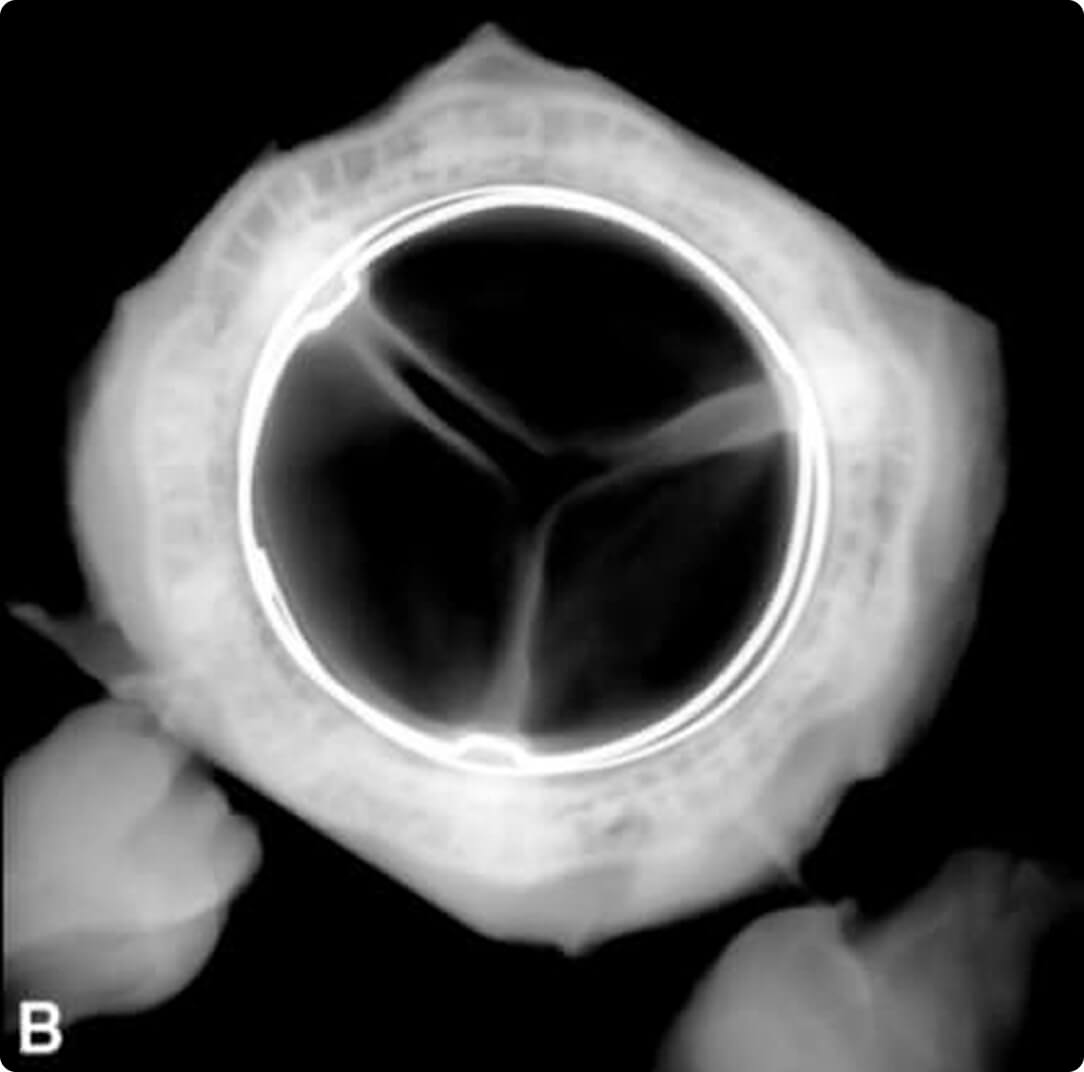
B = RESILIA Tissue Valve
This novel tissue preservation technology, when applied to the PERIMOUNT mitral valve, significantly improved hemodynamic and anti-calcification properties vs the standard PERIMOUNT valve.
References
- Bartus K, Litwinowicz R, Bilewska A, et al. Final 5-year outcomes following aortic valve replacement with a RESILIA™ tissue bioprosthesis. Eur J Cardiothorac Surg. 2021;59(2):434-441. doi:10.1093/ejcts/ezaa311
- Tod TJ, Gohres RA, Torky M, et al. Influence of Tissue Technology on Pannus Formation on Bioprosthetic Heart Valves. Cardiovasc Eng Technol. 2021;12(4):418-425. doi:10.1007/s13239-021-00530-1
- Flameng W, Hermans H, Verbeken E, Meuris B. A randomized assessment of an advanced tissue preservation technology in the juvenile sheep model. J Thorac Cardiovasc Surg. 2015;149(1):340-345. doi:10.1016/j.jtcvs.2014.09.062
Medical device for professional use.
For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
PP--EU-1509 v1.0
