
people in Europe have clinically relevant TR5

Medications may treat symptoms but not the TR itself, which can continue to progress.3,4

people in Europe have clinically relevant TR5

of patients with mild or trivial TR progressed to moderate or severe TR in ~3 years6‡

of patients with severe TR are estimated to die within 1 year of diagnosis2,7
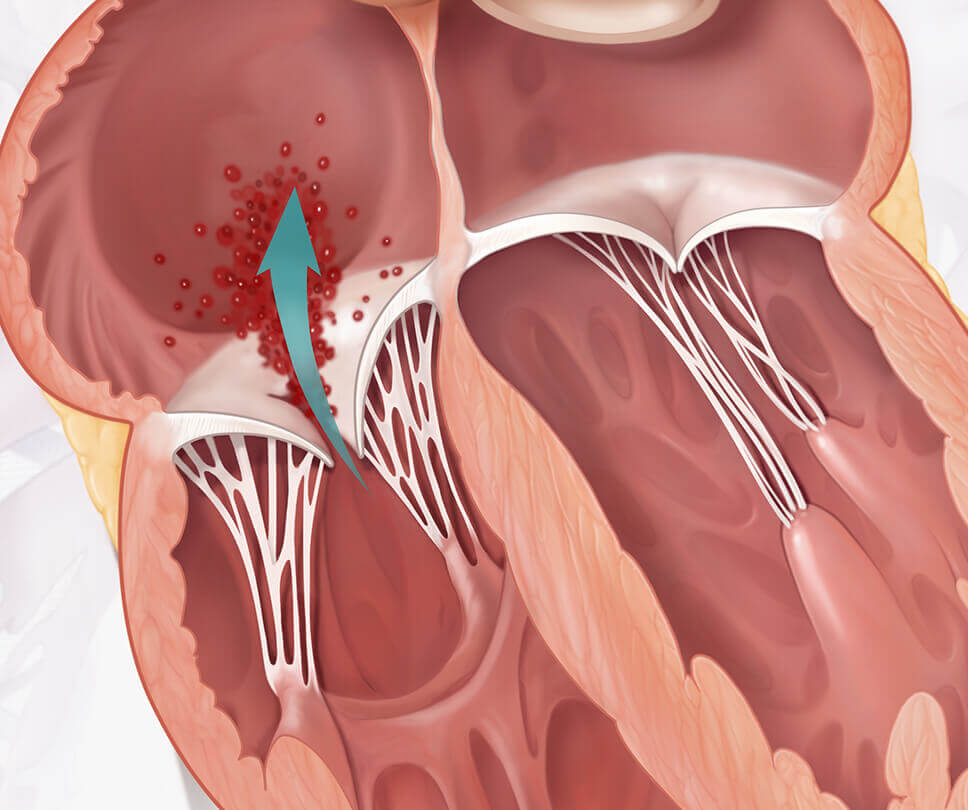
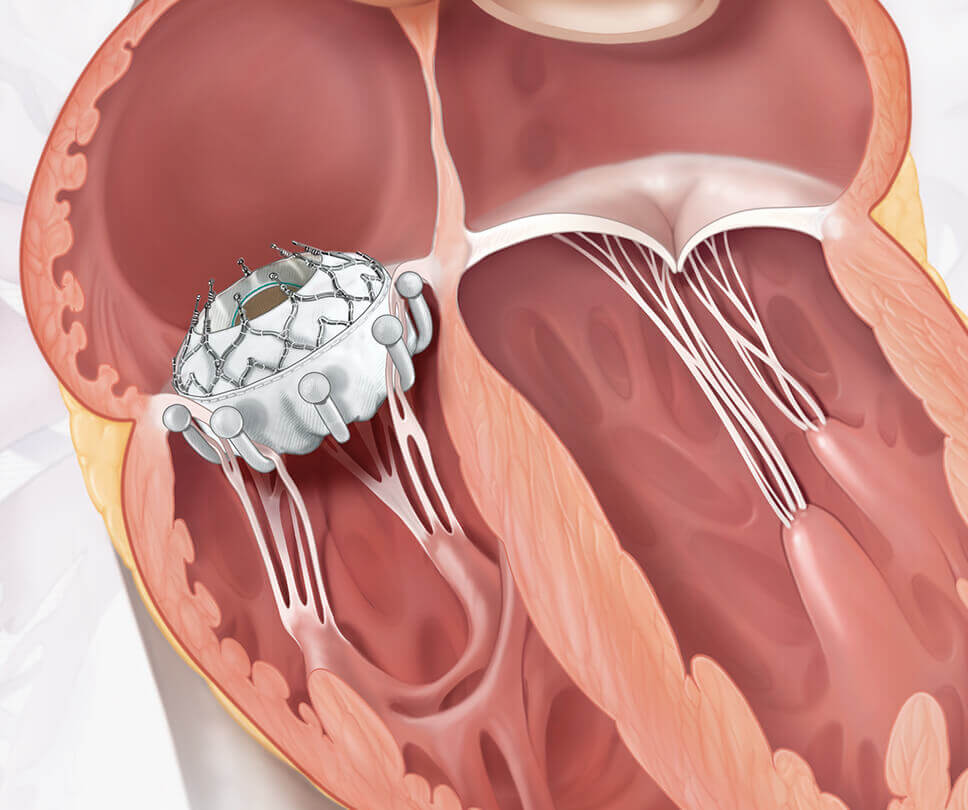
Move the arrow left or right to see the comparison between a heart with tricuspid regurgitation and one with the EVOQUE valve


Because TR can progress in severity, reducing TR severity may improve patient quality of life3,8
*First-of-its-kind device commercially available (in the EU).
†Based on US data.
‡Based on a retrospective echocardiographic analysis of Israeli patients.
§In a paired analysis in the TRISCEND II trial, 72.6% of EVOQUE TTVR patients achieved TR reduction to none/trace at 1 year follow-up (N=212).
The first-of-its-kind transcatheter tricuspid replacement option for eligible patients*
*First of its-kind-device commercially available (in the EU)
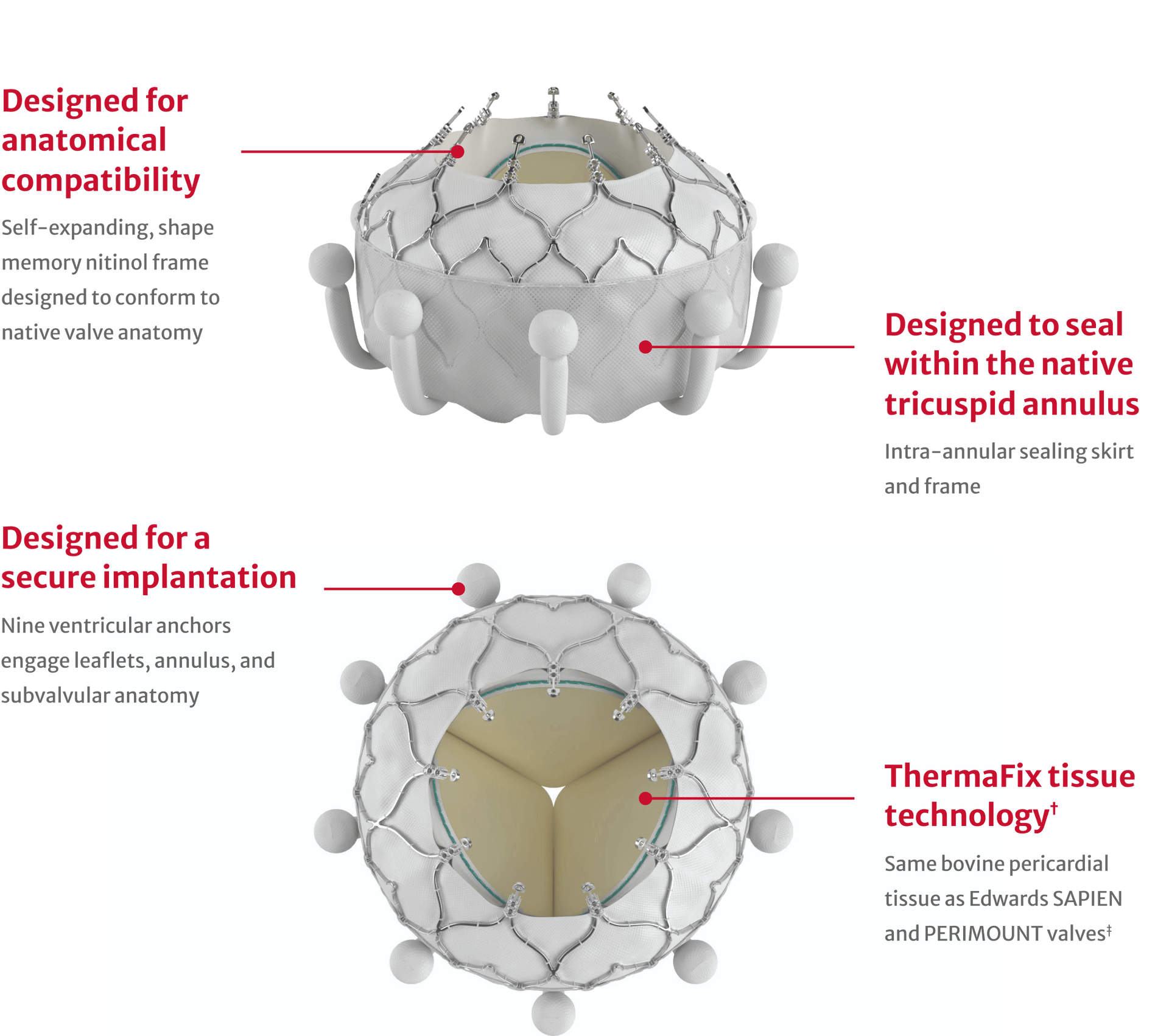
†No clinical data are available that evaluate the long-term impact of the Carpentier-Edwards ThermaFix tissue process in patients.
‡Excluding Edwards SAPIEN 3 Ultra RESILIA valves.

Procedural efficiency with an average device time of approximately one hour9

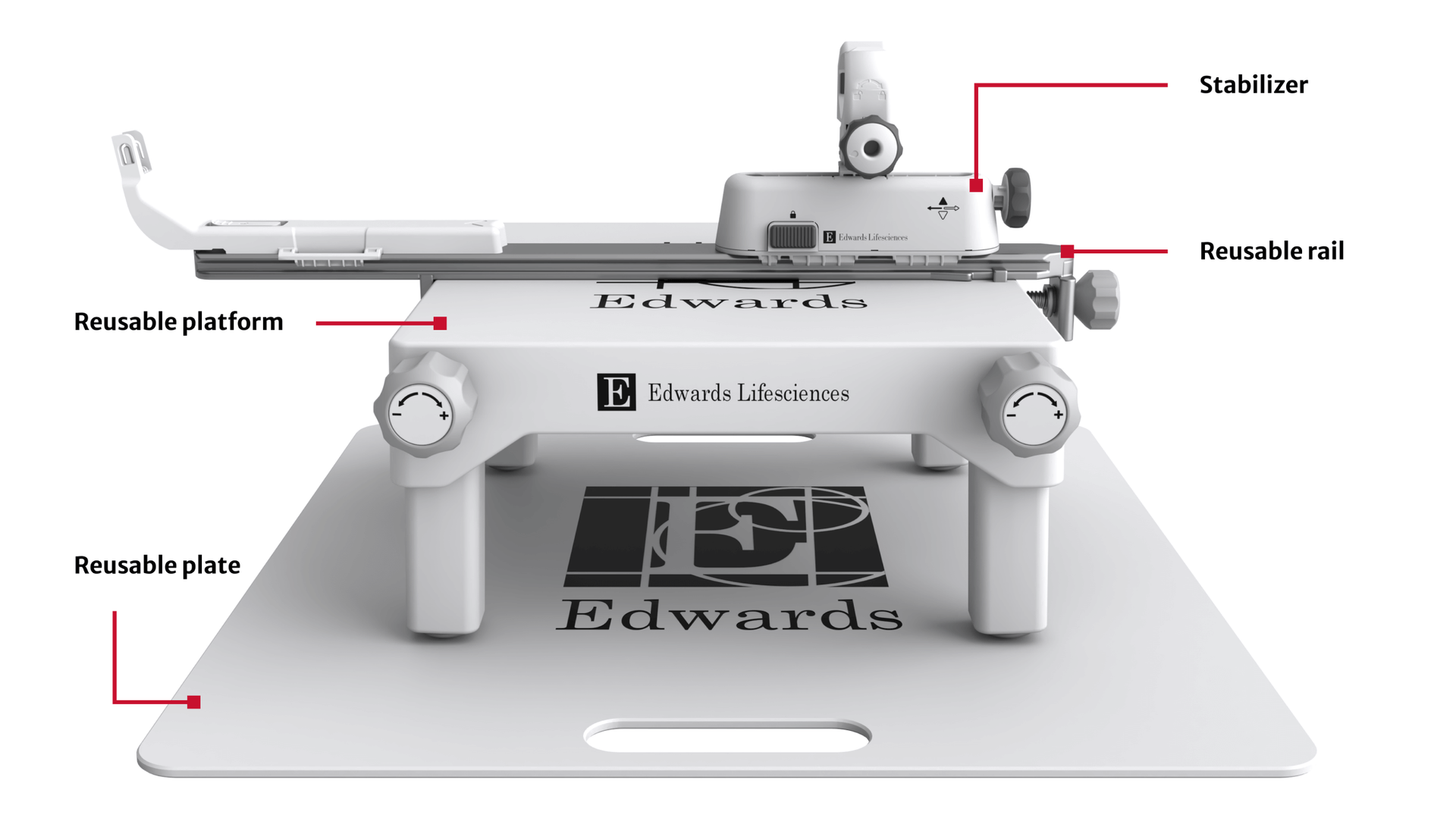
Low profile delivery system with 3 planes of movement designed for controlled positioning
Superior clinical benefit and consistent TR resolution for patients with EVOQUE TTVR

The TRISCEND II trial is a prospective multi-center randomized controlled trial evaluating the safety and effectiveness of transcatheter tricuspid valve replacement with the EVOQUE system plus optimal medical therapy compared to optimal medical therapy alone in patients with symptomatic severe ≥ TR. These data pertain to the entire cohort of 400 patients at 1 year.
The TRISCEND II trial successfully met the composite primary safety and effectiveness endpoint at 1 year.
Greater likelihood of clinical benefit with EVOQUE TTVR vs medical therapy alone
(95% CI, 1.56 to 2.62; p<0.001)

Greater likelihood of clinical benefit with EVOQUE TTVR vs medical therapy alone
(95% CI, 1.56 to 2.62; p<0.001)


Composite Endpoint

Win Ratio Analysis
Greater likelihood of clinical benefit with EVOQUE TTVR vs medical therapy alone
(95% CI, 1.56 to 2.62; p<0.001)
Greater likelihood of clinical benefit with EVOQUE TTVR vs medical therapy alone
(95% CI, 1.56 to 2.62; p<0.001)
≤mild TR with EVOQUE TTVR at 1 year
median device time




TR Reduction

Treats a diverse patient population

Treats a broad patient population
≤mild TR with EVOQUE TTVR at 1 year
median device time
*Refer to Instructions for Use.
†Refer to the Instructions for Use for indications, warnings, potential adverse events, and clinical experience associated with EVOQUE valve implantation in patients with pre-existing pacemakers.
Favorable trends observed in all-cause mortality and heart failure hospitalizations at 1 year between EVOQUE TTVR and medical therapy alone.
with EVOQUE TTVR vs 15.2% with medical therapy alone

with EVOQUE TTVR vs 26.1% with medical therapy alone

Cardiovascular mortality
Stroke
Severe bleeding
New pacemaker implant in pacemaker-naïve patients


Kaplan-Meier All-Cause Mortality

Kaplan-Meier Heart Failure Hospitalization

30-day Safety profile
with EVOQUE TTVR vs 15.2% with medical therapy alone
with EVOQUE TTVR vs 26.1% with medical therapy alone
Cardiovascular mortality
Stroke
Severe bleeding
New pacemaker implant in pacemaker-naïve patients
At 1-year follow-up compared to baseline, patients treated with EVOQUE TTVR experienced meaningful and sustained TR improvements in quality-of-life, functional status, and exercise capacity.
improvement in KCCQ-OS with EVOQUE TTVR at 1 year compared to baseline

of EVOQUE TTVR patients in NYHA Class I/II at 1 year

improvement in 6MWD with EVOQUE TTVR at 1 year compared to baseline


KCCQ-OS improvement

NYHA Class improvement

6MWD improvement
improvement in KCCQ-OS with EVOQUE TTVR at 1 year compared to baseline
of EVOQUE TTVR patients in NYHA Class I/II at 1 year
improvement in 6MWD with EVOQUE TTVR at 1 year compared to baseline
For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).